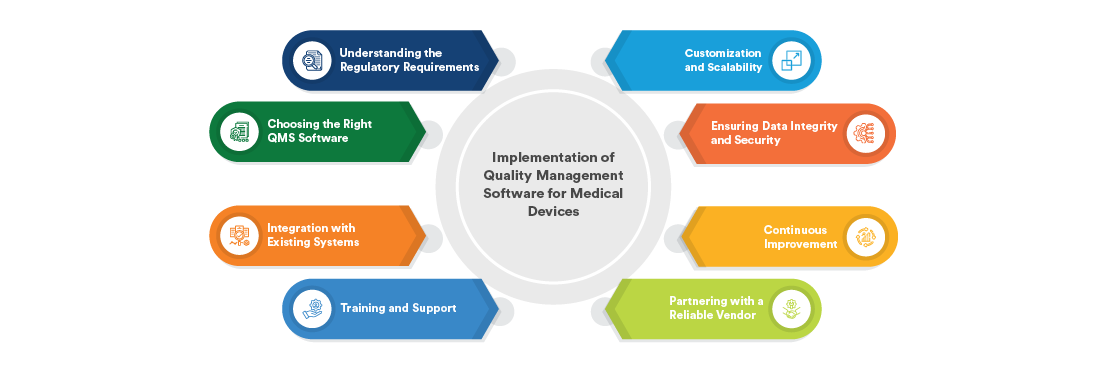
The medical equipment industry is one of the most sensitive and highly regulated areas worldwide. Unlike many other products, medical equipment directly affect human health and safety, which makes quality management more important than ever. To meet regulatory expectations and to build confidence with patients, healthcare providers and officials, organizations will have to apply a QMS for medical devices (MDQMS).
In ISO Gujarat, we specialize in helping manufacturers, suppliers and medical devices distributors and apply effective MDQMS framework that align with global standards and regulatory requirements.
What is QMS for Medical Devices (MDQMS)?
A QMS for medical devices, often referred to as MDQMS, is a structured system that defines process, procedures and responsibilities to ensure that medical equipment constantly meets security, quality and regulatory requirements.
This product covers every stage of the life cycle – from design and development to manufacturing, storage, distribution, installation and servicing. By adopting MDQMS, medical device companies ISO 13485, U.S. The FDA 21 CFR Part 820, and the European Union Medical Device can ensure compliance with international rules such as Regulation (EU MDR).
Why MDQMS is Important for Medical Equipment Industry
Bate is more in the medical equipment field. Even a minor defect or incompatibility can result in life-threatening results. This is the reason that regulators around the world gave companies a mandate to maintain QMS for medical equipment.
The main reasons for MDQMS include:
- Safety – To protect patient safety-end-users from risks and ensure devices reliability.
- Regulatory compliance – to meet global standards and legal requirements.
- Stability – Produced equipment that meet specifications without variations.
- Risk management – identifying, evaluating and controlling risks.
- Global Market Access – Certification allows entry into regulated markets worldwide.
In ISO Gujarat, we help to align businesses with these requirements to ensure both compliance and trust.
Major principles of MDQMS
An effective QMS for medical devices is based on the principles that integrate quality in every aspect of the product life cycle:
- Customer and patient focus – ensuring that equipment meets user needs and safety expectations.
- Leadership Commitment – Driving accountability and compliance from above.
- Risk-based approach – designing and managing risk during production.
- Regulatory Alignment – Compliance with local and international laws.
- Process efficiency – Minimizing errors, delays and reducing costs.
- Continuous improvement – customizing the system to develop technology and rules.
Regulatory Standards for QMS for Medical Devices
Many international standards direct the implementation of MDQMS:
- ISO 13485 – The most widely recognized international standard for QMS in medical devices.
- US FDA 21 CFR Part 820 – Quality System Regulation (QSR) for marketed equipment in the United States.
- EU MDR / IVDR – European Association regulated for medical and in-vitro diagnostic devices.
- Indian MDR 2017 – Regulatory framework controlling medical equipment in India.
ISO Gujarat supports organizations in understanding and implementing these standards.
Components of QMS for Medical Devices
An effective MDQMS contains the following main elements:
- Quality policy and purpose – commitment to safety and compliance.
- Design and development control – ensuring safe and effective product design.
- Documents and Record Control – Management of Quality Manual, SOP and Technical Files
- Suppliers Management – Evaluation and control suppliers to ensure compliance.
- Risk management – conducting risk analysis in life cycle.
- Production and process control – validation of manufacturing processes for stability.
- Corrective and preventive actions (CAPA)-Identification and addressing non-originated.
- Complaint dealing with – to effectively manage response and adverse events.
- Internal Audit – Ensuring ongoing compliance and readiness for external audit.
In ISO Gujarat, we tailor these components to correspond to the size, structure and product type of each organization.
Benefits of Implementing MDQMS
A strong QMS for medical devices provides long -term benefits:
- Better patient safety – reduces risk and the product increases reliability.
- Regulatory compliance – ensures alignment with national and international laws.
- Market Access – Certification opens the doors of global markets.
- Operations efficiency – streamlined procedures reduce costs and waste.
- Customer Trust – creates confidence among patients, doctors and healthcare providers.
- Low memorial and liabilities-product failures or low risk of non-transportation.
ISO Gujarat ensures that the business remains obedient and unlock these benefits.
MDQMS Implementation Process
A structured approach to applying a QMS for medical devices contains:
- Gap analysis – assessing existing processes against regulatory requirements.
- System Design – Creating policies, procedures and documentation.
- Training and awareness – construction capacity between employees.
- Process implementation – Implementing QMs in departments and operations.
- Internal Audit and Review – Evaluation of System Effectiveness and Compliance.
- Certification Preparation – Ensuring readiness for external audit and certificate
The ISO Gujarat offer end-to-end guidance, making MDQMS implementation a smooth process.
QMS for Medical Equipment in India
In India, with the increasing demand for both domestic and export markets, the medical equipment industry is growing rapidly. Medical device rules (MDR) 2017, implemented by Central Drugs Standard Control Organization (CDSCO), the mandate is that manufacturers comply with stringent quality requirements.
MDQMS is no longer optional, but there is a legal requirement for many categories tools. ISO Gujarat assisted Indian manufacturers to align with international standards such as MDR 2017 as well as ISO 13485, which ensures global recognition and compliance.
Why choose ISO Gujarat for MDQMS?
Medical equipment requires specialization in quality management and regulatory matters to apply QMS. In ISO Gujarat, we bring:
- Expert Guidance – Years of experience in ISO certification and medical equipment compliance.
- Customized solutions – Specific system for startups, SMEs and large manufacturers.
- End-to-end support – From documentation to training and certification.
- Regulatory Knowledge – Specialization in Indian MDR, FDA and European Union MDR rules.
- Continuous compliance – Support for ongoing audit, renewal and system updates.
Our goal is to ensure that your organization not only meets compliance requirements, but also creates a reputation for safety and reliability in the global market.